Publications
J. T. Calhoun, S. M. K. McKinnie
Chem Catal., 2026, 1, 101632
Invited preview of Bushin et al's Nat. Biotechnol. article: Growth-coupled biosynthesis of the animal pigment xanthommatin

J. L. Cordoza*, R. A. Johnson*, A. E. Whiteley, C. A. Horta, J. C. Nicholas, D. M. Owen, L. Rodriguez-Velandia, G. T. Luu, V. Z. Petukhova, J. T. Calhoun, L. M. Sanchez^, S. M. K. McKinnie^, K. R. Watts^
(*authors contributed equally, ^co-corresponding authors)
ACS Chem. Biol., 2026, accepted doi: 10.1021/acschembio.5c00877

A. R. Hopiavuori, B. S. Andre*, E. D. Avalos*, S. L. Beal*, G. R. Beck*, C. J. Choi*, E. M. Dolzhansky*, M. X. Du*, I. S. Gallardo*, C. J. Ghorbani*, K. M. Hinaga*, A. T. Nguyen*, S. M. K. McKinnie
(*authors contributed equally)
J. Nat. Prod., 2026, 89, 198-207.
selected as the ACS Editor's Choice article for Dec. 30, 2025!
highlighted on UCSC Teaching and Learning Center's Resources page!

39) A high-throughput biocatalytic platform for screening isomeric kainoid natural products
R. A. Shepherd*, M. Ramachandra*, A. R. Hopiavuori, M. C. Jones, C. A. Fihn, A. J. Tabag, A. Manjunath, L. Y. Gad, C. R. Whipple, S. M. K. McKinnie^, L. M. Sanchez^
(*authors contributed equally, ^co-corresponding authors)
Cell Rep. Phys. Sci., 2026, 103092. doi: 10.1016/j.xcrp.2025.103092
highlighted by UCSC Newsroom and EurekAlert!

J. T. Baumgartner, L. A. Varga, J. T. Calhoun, V. H. Balasco Serrão, S. Loerch^, S. M. K. McKinnie^
(^co-corresponding authors)
submitted, 2025, bioRxiv: doi: 10.1101/2025.10.24/684477

N. K. Krull*, M. Strobel*, J. Saulog, L. Zaroubi, B. S. Paulo, M. Timba, D. R. Braun, G. Mingolelli, J. Raherisoanjato, R. A. Shepherd, A. F. Scott, C. De Silva, C. Fergusson, Z. Daniel, S. K. Pokharel, S. Romanowski, A. Hernandez, M. Monge-Loría, C. E. Dylla, M. M. Natu, V. Z. Petukhova, N. Garg, P. R. Jensen, A. Blachowicz, C. D. Cassilly, L. Guan, D. C. Stevens, J. M. Winter, S. M. K. McKinnie, B. I. Adaikpoh, S. Carlson, E. P. McCauley, W. W. Metcalf, T. S. Bugni, M. W. Mullowney, E. G. Pamer, M. T. Henke, H. Barton, D. O. Carter, A. S. Eustáquio, R. G. Linington, L. M. Sanchez^, M. Wang^, B. T. Murphy^
(*authors contributed equally, ^co-corresponding authors)
submitted, 2025, bioRxiv: doi: 10.1101/2025.10.15.682631

R. A. Shepherd, C. A. Fihn, A. J. Tabag, S. M. K. McKinnie, L. M. Sanchez
Nat. Prod. Rep., 2025, 42, 1037-1054.

J. T. Baumgartner, C. S. McCaughey, H. S. Fleming, A. R. Lentz, L. M. Sanchez, S. M. K. McKinnie
2024, bioRxiv: doi: 10.1101/2024.07.31.606109

34) Regioselective halogenation of lavanducyanin by a site-selective vanadium-dependent chloroperoxidase
J. T. Baumgartner, S. M. K. McKinnie
Org. Lett., 2024, 26, 5725-5730.

A. R. Hopiavuori*, R. T. Huffman*, S. M. K. McKinnie
(*authors contributed equally)
Methods Enzymol., 2024, 704, 233-258.

32) Vanadium haloperoxidases as noncanonical terpene synthases
J. T. Baumgartner*, L. I. Lozano Salazar*, L. A. Varga, G. H. Lefebre, S. M. K. McKinnie
(*authors contributed equally)
Methods Enzymol., 2024, 699, 447-475.

31) Algal kainoid synthases exhibit substrate-dependent hydroxylation and cyclization activities
A. R. Hopiavuori, S. M. K. McKinnie
ACS Chem. Biol., 2023,18, 2457-2463.

J. L. Cordoza, P. Y.-T. Chen, L. R. Blaustein, S. T. Lima, M. F. Fiore, J. R. Chekan, B. S. Moore, S. M. K. McKinnie
ACS Catal., 2023, 13, 9817-9828.

29) Biosynthesis of guanitoxin enables global environmental detection in freshwater cyanobacteria
S. T. Lima, T. R. Fallon*, J. L. Cordoza*, J. R. Chekan, E. Delbaje, A. R. Hopiavuori, D. O. Alvarenga, S. M. Wood, H. Luhavaya, J. T. Baumgartner, F. A. Dörr, A. Etchegaray, E. Pinto, S. M. K. McKinnie^, M. F. Fiore^, B. S. Moore^.
(*authors contributed equally, ^co-corresponding authors)
J. Am. Chem. Soc., 2022, 114, 9372-9379.

J. T. Baumgartner, S. M. K. McKinnie.
mSystems, 2021, 6, e00780-21.

P. M. O. Pimentel, D. R. R. de Assis, M. Gualdrón-Lopez, A. Barroso, F. Brant, P. G. Leite, B. C. L. Oliveira, L. Esper, S. M. K. McKinnie, J. C. Vederas, M. N. Cordeiro, P. V. M. dos Reis, M. M. Teixeira, A. M. C. Pimenta, M. H. Borges, M. E. de Lima, F. S. Machado
Clin. Immunol., 2021, 226, 108713.

L. A. M. Murray, S. M. K. McKinnie, B. S. Moore, J. H. George.
Nat. Prod. Rep., 2020, 37, 1334-1366.

D. P. Goméz-Mendoza, R. P. Lemos, I. C. G. Jesus, V. Gorshkov, S. M. K. McKinnie, J. C. Vederas, F. Kjeldsen, S. Guatimosim, R. A. Santos, A. M. C. Pimenta, T. Verano-Braga.
J. Proteome Res., 2020, 19, 3467-3477.

24) Comparative genomics and metabolomics in the genus Nocardia
D. Männle, S. M. K. McKinnie, S. S. Mantri, K. Steinke, Z. Lu, B. S. Moore, N. Ziemert, L. Kaysser.
mSystems, 2020, 5, e00125-20.

J. R. Chekan, S. M. K. McKinnie, J. P. Noel, B. S. Moore.
Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 12799-12805.

22) Nonlinear biosynthetic assembly of alpiniamide by a hybrid cis/trans-AT PKS-NRPS
R. Sigrist, H. Luhavaya, S. M. K. McKinnie, A. F. da Silva, I. D. Jurberg, B. S. Moore, L. G. de Oliveira.
ACS Chem. Biol., 2020, 15, 1067-1077.

21) Guanitoxin, re-naming a cyanobacterial organophosphate toxin
M. F. Fiore, S. T. de Lima, W. W. Carmichael, S. M. K. McKinnie, J. R. Chekan, B. S. Moore.
Harmful Algae, 2020, 92, 101737.

Publications before UCSC
X. Tang, Y. Kudo, J. L. Baker, S. LaBonte, P. A. Jordan, S. M. K. McKinnie, J. Guo, T. Huan, B. S. Moore, and A. Edlund.
ACS Infect. Dis., 2020, 6, 563-571.
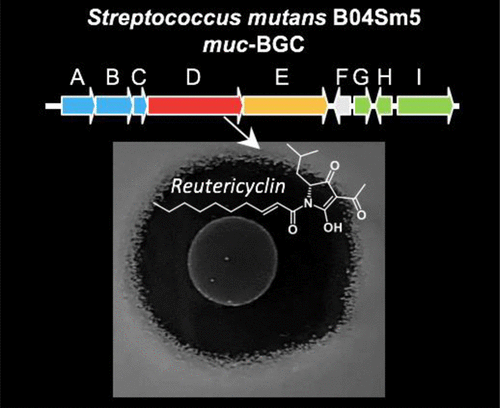
19) Macrocyclic colibactin induces DNA double-strand breaks via copper-mediated oxidative cleavage
Z.-R. Li, J. Li, W. Cai, J. Y. H. Lai, S. M. K. McKinnie, W.-P. Zhang, B. S. Moore, W. Zhang, and P.-Y. Qian.
Nat. Chem., 2019, 11, 880-889.

18) Scalable biosynthesis of the seaweed neurochemical kainic acid.
J. R. Chekan, S. M. K. McKinnie, M. L. Moore, S. G. Poplawski, T. P. Michael, and B. S. Moore.
Angew. Chem. Int. Ed., 2019, 58, 8454-8457.

H. L. Luhavaya, R. Sigrist, J. R. Chekan, S. M. K. McKinnie, and B. S. Moore.
Angew. Chem. Int. Ed., 2019, 58, 8394-8399.

M. L. Rothe, J. Li, E. Garibay, B. S. Moore*, and S. M. K. McKinnie*.
(*co-corresponding authors)
Org. Biomol. Chem., 2019, 17, 3416-3423.

C. Fischer, T. Lamer, W. Wang, S. M. K. McKinnie, X. Iturrioz, C. Llorens-Cortes, G. Y. Oudit, and J. C. Vederas.
Eur. J. Med. Chem., 2019, 166, 119-124.

14) Total enzyme syntheses of napyradiomycins A1 and B1.
S. M. K. McKinnie*, Z. D. Miles*, P. A. Jordan, T. Awakawa, H. P. Pepper, L. A. M. Murray, J. H. George, and B. S. Moore.
(*authors contributed equally)
J. Am. Chem. Soc., 2018, 140, 17840-17845.

13) Biosynthesis of the neurotoxin domoic acid in a bloom-forming diatom.
J. K. Brunson*, S. M. K. McKinnie*, J. R. Chekan, J. P. McCrow, Z. D. Miles, E. M. Bertrand, V. A. Bielinski, H. Luhavaya, M. Oborník, G. J. Smith, D. A. Hutchins, A. E. Allen, and B. S. Moore. (*authors contributed equally)
Science, 2018, 361, 1356-1358.

L. A. M. Murray, S. M. K. McKinnie, H. P. Pepper, R. Erni, Z. D. Miles, M. C. Cruickshank, B. López-Pérez, B. S. Moore, and J. H. George.
Angew. Chem. Int. Ed. Engl., 2018, 57, 11009-11014.

11) Characterization and biochemical assays of Streptomyces vanadium-dependent chloroperoxidases.
S. M. K. McKinnie, Z. D. Miles, and B. S. Moore.
Methods Enzymol., 2018, 604, 405-424.

J. Zheng, S. M. K. McKinnie, A. E. El Gamal, W. Feng, Y. Dong, V. Agarwal, W. Fenical, A. Kumar, Z. Cao, B. S. Moore, and I. N. Pessah.
Environ. Sci. Technol., 2018, 52, 5469-5478.

S. M. K. McKinnie, W. Wang, C. Fischer, T. McDonald, K. R. Kalin, X. Iturrioz, C. Llorens-Cortes, G. Y. Oudit, and J. C. Vederas.
J. Med. Chem., 2017, 60, 6408-6427.

8) Targeting the apelin pathway as a novel therapeutic approach for cardiovascular disease.
J.-C. Zhong, Z.-Z. Zhang, W. Wang, S. M. K. McKinnie, J. C. Vederas, and G. Y. Oudit.
Biochem. Biophys. Acta, Mol. Basis Dis., 2017, 1863, 1942-1950.

7) The metalloprotease neprilysin degrades and inactivates apelin peptides.
S. M. K. McKinnie, C. Fischer, K. M. H. Tran, W. Wang, F. Mosquera, G. Y. Oudit, and J. C. Vederas.
ChemBioChem., 2016, 17, 1495-1498.

W. Wang, S. M. K. McKinnie, M. Farhan, M. Paul, T. McDonald, B. McLean, C. Llorens-Cortes, S. Hazra, A. G. Murray, J. C. Vederas, and G. Y. Oudit.
Hypertension, 2016, 68, 365-377.

5) Differential response of orthologous L,L-diaminopimelate aminotransferases (DapL) to enzyme inhibitory antibiotic lead compounds.
S. M. K. McKinnie, E. M. Rodriguez-Lopez, J. C. Vederas, J. M. Crowther, H. Suzuki, R. C. J. Dobson, T. Leustek, A. J. Triassi, M. S. Wheatley, and A. O. Hudson.
Bioorg. Med. Chem., 2014, 22, 523-530.

W. Wang, S. M. K. McKinnie, V. B. Patel, G. Haddad, Z. Wang, P. Zhabyeyev, S. K. Das, R. Basu, B. McLean, V. Kandalam, J. M. Penninger, Z. Kassiri, J. C. Vederas, A. G. Murray, and G. Y. Oudit.
J. Am. Heart Assoc., 2013, 2, e000249.
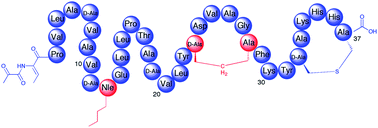
3) The solid phase supported peptide synthesis of analogues of the lantibiotic lactocin S.
S. M. K. McKinnie, A. C. Ross, M. J. Little, and J. C. Vederas.
MedChemComm, 2012, 3, 971-975.
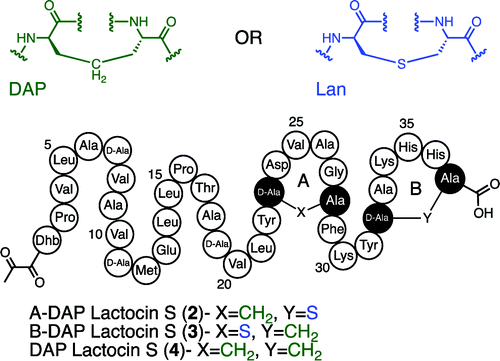
2) The synthesis of active and stable diaminopimelate analogues of the lantibiotic peptide lactocin S.
A. C. Ross, S. M. K. McKinnie, and J. C. Vederas.
J. Am. Chem. Soc., 2012, 134, 2008-2011.
V. R. Pattabiraman, S. M. K. McKinnie, and J. C. Vederas.
Angew. Chem. Int. Ed., 2008, 47, 9472-9475.
